ACCREDITATION STATEMENT
This activity has been planned and implemented by Ciné-Med and the Angiogenesis Foundation. Ciné-Med is accredited by the American Nurses Credentialing Center (ANCC), the Accreditation Council for Pharmacy Education (ACPE), and the Accreditation Council for Continuing Medical Education (ACCME), to provide continuing medical education for the health care team.
CREDIT DESIGNATION
Ciné-Med designates this enduring material for a maximum of 1.5 AMA PRA Category 1 Credits™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Credit will be awarded to those who complete this activity according to the instructions and achieve a score of 70% or better on the post activity examination. A certificate of credit will be available for download to those who successfully complete the examination.
RELEASE AND EXPIRATION
Date of original release: June 11, 2014
Date of expiration: June 10, 2015
CME Course Code: 2014OVARIAN
TARGET AUDIENCE
Practicing oncologists and primary care physicians in the U.S., researchers and medical students
HEALTHCARE GAP
In the United States alone, more than 22,240 new cases of ovarian cancer are estimated to have been diagnosed in 2013, and an estimated 14,230 will have died from this disease. Ovarian cancer is the ninth most common cancer among women, excluding non-melanoma skin cancers. It ranks fifth in cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system. Ovarian cancer accounts for about 3% of all cancers in women. A woman’s risk of getting ovarian cancer during her lifetime is about 1 in 72. Her lifetime chance of dying from ovarian cancer is about 1 in 100.
PROGRAM LEARNING OBJECTIVES
At the completion of this activity, participants should be able to:
- Summarize the current clinical progress of targeted therapies in the management of ovarian cancer in early and late stage clinical trials.
- Explain current clinical challenges in the management of ovarian cancer.
- Interpret the outcome data from recent well-designed scientific and clinical studies of protocols studying new-targeted therapies.
- Integrate side effect management into the long term management of patients with ovarian cancer.
- Assess treatment options, efficacy data, and side effect management with members of the cancer treatment team, as well as cancer patients and their family members.
ACTIVITY GOAL
This activity is designed to address the following ABMS / IOM competencies:
Patient Care and Medical Knowledge
METHOD OF PARTICIPATION
There are no fees for participating in and receiving credit for this online educational activity. The participant should, in order, read the objectives and faculty disclosures, review the educational content, answer the multiple-choice post-test and complete the evaluation. This program is available in PDF format accessible from the Angiogenesis Foundation’s website (http://www.angio.org) in the CME section. A print version is also available; for more information contact outreach@angio.org. After reviewing the material, CME credits are available through the Angiogenesis Foundation’s website (http://www.cmeonline.org) by selecting the name of the program (registration required). Course code: 2014OVARIAN
ACKNOWLEDGEMENT OF SUPPORT
This activity is supported by educational grants from Genentech.
COURSE FACULTY
William Li, MD
Dartmouth Medical College
DISCLOSURE
Cine-Med adheres to accreditation requirements regarding industry support of continuing medical education. Disclosure of the planning committee and faculty’s commercial relationships will be made known at the activity. Speakers are required to openly disclose any limitations of data and/or any discussion of any off-label, experimental, or investigational uses of drugs or devices in their presentations.
All Cine-Med and Angiogenesis Foundation employees in control of content have indicated that they have no relevant financial relationships to disclose.
William W. Li, M.D.
President, the Angiogenesis Foundation
Editor-in-Chief
Disclosure: No relevant financial relationships to disclose
TOPICS AND EDUCATIONAL CONTENT
The State of Antiangiogenic Therapy for Advanced Ovarian Cancer:
- The State of Antiangiogenic Therapy for Advanced Ovarian Cancer
- Anti-VEGF Monoclonal Antibody
- Multitargeted Antiangiogenic Agents
- Metronomic Chemotherapy
- Biomarkers
- Safety of Antiangiogenic Therapy for Ovarian Cancer
- Future Directions
SYSTEM REQUIREMENTS
This educational program is available as a mobile application and a PDF file. The mobile application can be run on any iOS or Android device. To view and print PDF files, you must have Adobe Reader installed on your computer. Most computers already have this software installed. If yours does not, you can download Adobe Reader free from the Adobe Web site: http://www.adobe.com.
For questions about this program, please contact the Angiogenesis Foundation at 617-401-2779 or outreach@angio.org.
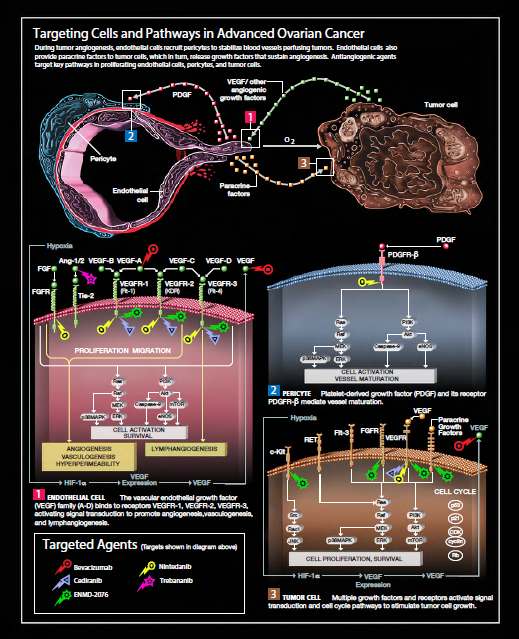
TARGETING TUMOR ANGIOGENESIS
Epithelial ovarian cancer (EOC), along with primary peritoneal cancer (PPC), is the fifth leading cause of cancer deaths in women in the United States, with an estimated 14,270 deaths and 21,980 new cases diagnosed in 2014.(1) Localized disease accounts for approximately 15% of the women diagnosed with ovarian cancer, since they are often asymptomatic until the disease is very advanced; the 5-year survival rate for the women with this type of disease is 92%.(2) On the other hand, 61% of women diagnosed with ovarian cancer already have distant metastases, and the associated 5-year survival rate for them is only 27%.(3) Standard therapy for advanced disease has traditionally relied on cytotoxic agents, such as DNA platinating agents, nucleoside analogs, and taxanes, all which kill rapidly dividing tumor cells by disrupting DNA or the microtubule integrity. Despite the efficacy of these cytotoxic agents, the majority of women still experience relapse and subsequently progress to treatment-refractory disease.
Antiangiogenic agents belong to a class of drugs that has been used as treatment for various tumor types. The antitumor activity of these agents is based on their ability to interrupt or mimic cell signals at the molecular level and interfere with tumor angiogenesis and survival.(4) One primary mediator of tumor angiogenesis is the substance vascular endothelial growth factor (VEGF), which induces the proliferation and migration of endothelial cells and increases vascular permeability.(5) The presence of hypoxia in the cellular microenvironment enhances the expression of VEGF in tumor cells and leads to unregulated new vessel growth.(6) Unlike the vessels found in normal tissues, tumor vasculature is permeable and highly disorganized, which propagates tumor hypoxia and growth factor expression, and can potentially interfere with therapeutic drug delivery.
Antiangiogenic agents function by disrupting the proangiogenic growth factor communication loop between tumor cells and resident endothelial cells (Please refer to the illustration on page 4). Other substances besides VEGF, such as fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), insulin-like growth factor (IGF), and hepatocyte growth factor (HGF) can also increase endothelial cell proliferation after binding with their respective receptors. Additionally, angiotensin-1 influences endothelial cell survival by suppressing apoptosis when VEGF is present, and by inducing apoptosis in its absence.(7) When VEGF binds to its receptor, the vascular endothelial growth factor receptor (VEGFR), on the surface of endothelial cells, receptor dimerization leads to the phosphorylation of key tyrosine residues and the activation of numerous downstream signaling pathways.(8,9) Two of these pathways that play an important role in endothelial and tumor cell growth and survival are the Raf-MEK-Erk and phosphotidylinositol- 3-kinase(PI3K)- Akt pathways.(10) Each factor and process in these proangiogenic pathways represents potential targets for drug development and mechanisms of action for the treatment of cancer.
Numerous therapeutic agents that inhibit angiogenesis have been evaluated for activity in the treatment of advanced ovarian cancer. Angiogenesis plays an important role in both the cyclical nature of normal ovarian function and ovarian tumorigenesis.(11) Research has shown that VEGF expression is increased in ovarian tumors compared to normal ovaries, and that high VEGF expression in tumors has been linked to poorer prognosis.(12,13) The effects of VEGF expression on vascular permeability may also be a significant contributor to the development of malignant ascites in ovarian cancer patients.14,15 It follows that VEGF inhibition in animal models of ovarian cancer significantly modifies tumor vasculature, inhibits ascitic fluid accumulation, and decreases tumor growth.16
For more than ten years, multiple antiangiogenic agents with different mechanisms of action have been evaluated for use in the treatment of patients with ovarian cancer. This review will update clinical trial data regarding the use antiangiogenic agents for the treatment of platinum-sensitive and platinum-resistant advanced ovarian cancer.
Anti-VEGF Monoclonal Antibody: Bevacizumab
Bevacizumab is a humanized anti-VEGF monoclonal antibody that specifically binds VEGF in the extracellular space and prevents it from combining with VEGFR. Bevacizumab (BV) has been approved by the FDA for the treatment of metastatic colorectal cancer, non-squamous non-small cell lung cancer, glioblastoma, and advanced renal cell carcinoma, as monotherapy or as part of combination therapy.(17) The activity of BV in first-line therapy and in recurrent/platinum-sensitive and platinum-resistant disease has been evaluated in a number of phase 2 and 3 clinical trials.
Bevacizumab as First-Line Therapy
Several phase 2 trials have demonstrated that treatment with BV could elicit tumor response and delay disease progression.(18,19,20,21) As a result, two randomized phase 3 trials were conducted to evaluate the addition of BV to standard front-line chemotherapy for advanced ovarian cancer. One small study evaluated paclitaxel (175 mg/m2), carboplatin (AUC of 5 IV), and bevacizumab (15 mg/ kg of body weight) q21 days for six cycles in women with advancedstage ovarian cancer.(22) Of the 18 patients that were evaluated, there was an overall response rate (ORR) of 80%, with 30% having a complete response (CR). The activity of BV as an initial therapy was confirmed in an open-label phase 2 trial that investigated the addition of BV 15 mg/kg q3w to carboplatin AUC 5/paclitaxel 175 mg/m2.(23) The study participants were women who had with newly diagnosed advanced (stage ≥ IC) epithelial mullerian tumors; 79% of the women had optimally cytoreduced disease. Using combined RECIST and formal Rustin criteria, researchers reported an ORR of 76%, with CR in 21% of patients. The median progressionfree survival (mPFS) was 29.8 months, while median overall survival (mOS) had not yet been reached.
The phase 3 GOG 0218 trial was initiated to evaluate the superiority of adding BV to standard first-line therapy and to compare the effect of longer and shorter periods of VEGF suppression.(24) In this doubleblind and placebo-controlled trial, a total of 1,873 women with previously untreated advanced (stage III or IV), EOC, PPC, or fallopian tube cancer were randomized to receive 6 cycles of carboplatin AUC 5/paclitaxel 175 mg/m2 (chemotherapy), 6 cycles of chemotherapy plus BV 15 mg/kg q3w (BV-initiation), or 6 cycles of chemotherapy plus 22 cycles of BV 15 mg/kg q3w (BV-throughout). The median PFS was significantly prolonged in the BV-throughout arm when compared to the chemotherapy arm (14.1 months vs. 10.3 months, P < 0.001), but not in the BV-initiation arm (11.2 months vs. 10.3 months, P = 0.16).
The BV-associated improvement in PFS was seen for all prognostic groups. Median overall survival was similar for chemotherapy, BV-initiation, and the BV-throughout treatment groups (39.3, 38.7, and 39.7 months, respectively), and this was likely due to the confounding effects of post-progression treatment, particularly the crossover to antiangiogenic therapy in the management of recurrent disease.
The ICON 7 (International Cooperative Group for Ovarian Neoplasia) phase 3 trial investigated the addition of BV to carboplatin/paclitaxel as first-line therapy for advanced ovarian cancer, but at half the dose intensity used in GOG 0218.(25) A total of 1528 women diagnosed with high-risk early stage or advanced epithelial ovarian cancer, primary peritoneal cancer, or fallopian-tube cancer were randomized to receive either 6 cycles of carboplatin AUC 5/paclitaxel 175 mg/m2 (chemotherapy) or chemotherapy plus concurrent BV 7.5 mg/kg q3w (chemotherapy-BV) for 6 cycles followed by continued BV for 12 additional cycles. The addition of BV to chemotherapy improved median PFS by 1.7 months (19.8 months vs. 17.3, P = 0.004). The maximum benefit from BV treatment occurred at 12 months, which coincided with the end of BV exposure, and diminished thereafter such that PFS was slightly higher in the chemotherapy group by 24 months. The subgroup analysis showed that the addition of BV to chemotherapy greatly improved the median PFS (16.0 vs. 10.5 months, P = 0.002) in patients at high risk for progression. The updated analysis also revealed that this same subgroup of patients benefited from BV treatment in terms of median OS (36.6 vs. 28.8 months, P = 0.002) as well at this time.
The researchers also found that the mean global quality of life score at 54 weeks was higher in the standard chemotherapy group than in the BV group (P < 0.0001).(26) In 2013, the final overall survival results for ICON 7 showed that with a median follow-up 49 months, using Restricted Mean Survival Time (RMST), there was a nonsignificant improvement of 0.9 months in OS from 44.6 to 45.5 months with the addition of BV (P = 0.85).(27) In the high-risk group, there was an improvement of 4.8 months in RMST from 34.5 to 39.3 months (P = 0.03). Both the GOG 0218 and ICON7 demonstrated that adding BV to carboplatin and paclitaxel standard first-line therapy significantly prolonged PFS in women with advanced ovarian cancer, and the ICON7 subgroup analysis and overall survival results both revealed a significant BV-associated survival benefit for patients at high risk for progression.
Recurrent, Platinum-Sensitive Disease
Another phase 3 trial, OCEANS, was implemented to assess the efficacy and safety of BV in the treatment of patients with recurrent, platinum-sensitive advanced ovarian cancer.(28) Treatment of this patient population with combination therapy of bevacizumab with gemcitabine/carboplatin (GC) had been approved by the FDA in 2006 after the results of a European phase 3 intergroup trial demonstrated its superiority to gemcitabine alone.(29) Investigators initiated OCEANS in response to the results from a phase 2 trial, the GOG 170D study, which indicated that BV monotherapy had activity in treating patients with platinum-sensitive and platinum-refractory disease after recurrence.(30) Of the 62 women that were enrolled in this phase 2 study, 58% had a platinum-free interval (PFI) of less than 6 months and were thus considered platinum-sensitive. Investigators reported a 21% ORR, which included two CRs and 11 PRs. Median PFS and OS were 4.7 months and 17 months, respectively. Importantly, the investigators reported no significant association of prior platinum sensitivity or number of prior treatments with death, suggesting that BV had enduring activity in recurrent ovarian cancer.
The OCEANS trial, an industry-sponsored, placebo-controlled trial, randomized 484 women with platinum-sensitive ovarian cancer (PFI of at least 6 months) after first-line therapy to treatment with either GC AUC 4/1000 mg/m2 plus placebo (GC) or GC plus BV 15 mg/kg q3w (GC-BV).(31) The median PFS was 4 months longer in patients treated with GC-BV compared to those treated with GC (12.4 vs. 8.4 months, P < 0.0001). The addition of BV to GC was also associated with a statistically significant improvement in the ORR (78.5% vs. 57.4%, P < 0.0001), of which the majority were partial responses (148 of 242 and 117 of 242, respectively). The data with a high degree of censoring yielded a median OS of 35.2 months for GC-treated patients and 33.3 months for GC-BV-treated patients. In the analysis of the final survival results for this study presented in 2014, the median follow-up was 57.5 months among all patients.(32) The median OS was 32.9 months in the GC arm and was 33.6 months in the BV arm (HR: 0.952; 95% CI: 0.771–1.176). The median OS in both arms was considerably longer than the 18 months predicted based on prior data and used for the study design; the OS was also similar between the arms.
Together with the results of GOG 0218 and ICON7, the results from OCEANS have suggested that BV improved efficacy in terms of PFS of standard chemotherapy doublets for patients with chemotherapynaive platinum-pretreated ovarian cancer.
Platinum-Resistant/Refractory Disease
For women with advanced ovarian cancer, treatment in the platinum-resistant setting is an important one. Improving outcomes in these patients represents an unmet medical need; a high recurrence rate and typical survival times of less than 12 months are common in treatment-refractory disease. To determine whether BV could improve outcomes in this refractory patient population, the investigators initiated the industry-sponsored phase 3 study, AURELIA.(33) This randomized,(34) open-label phase 3 trial compared chemotherapy (investigator’s choice: paclitaxel 80 mg/m2 days 1, 8, 15, 22 q4w, topotecan 4 mg/m2 days 1, 8, 15 q4w, or liposomal doxorubicin 40 mg/m2 day 1 q4w) and chemotherapy plus BV 15 mg/kg q3w as salvage therapy in patients with advanced ovarian cancer. Patients were treated until progression or unacceptable toxicity, when they were crossed over to treatment with chemotherapy alone (after initial BV exposure) or BV monotherapy (after progression with initial chemotherapy alone). The results of the trial showed that the addition of BV to chemotherapy was associated with an increase in the median PFS (6.7 vs. 3.4 months, P < 0.001); this finding was consistent across all subgroups analyzed. Additionally, using RECIST criteria, more than twice as many patients treated with BV had an objective response (27.3% vs. 11.8%, P = 0.001) when compared to those treated with chemotherapy alone. For survival, the AURELIA authors found that the median OS was 13.3 months with chemotherapy compared with 16.6 months with BV and chemotherapy. This was not statistically significant. The authors did note that the trial was not intended to detect a difference in OS, because crossover to bevacizumab was permitted and had occurred in 40% of patients who were initially and randomly assigned to chemotherapy. The data from the AURELIA study, combined with the findings from the GOG-0218, ICON7, and OCEANS studies all provide evidence for the use of bevacizumab in the treatment of ovarian cancer.
Two earlier phase 2 trials.(35,36) had also evaluated the potential role of BV in the treatment of platinum-resistant ovarian cancer and led the way for the phase 3 analysis. In the first trial, patients were treated with albumin-bound paclitaxel 100 mg/m2 days 1, 8, and 15 and BV 10 mg/kg q2w until disease progression.(37) The overall response rate was 50% with four complete and 20 partial responses; the median PFS was 8.08 months and the median OS was 17.15 months. A second phase 2 study conducted in this disease setting assessed the activity of BV 10 mg/kg q2w in combination with topotecan (4 mg/m2 days 1, 8, 15 q4w) resulted in comparable measures of efficacy: median PFS was 7.8 months and median OS was 16.6 months.(38) Interestingly, this study suggested that patients who had undergone 2 previous treatment regimens received substantially greater benefit from BV than those who had only received one prior regimen; patients in this subgroup had significantly longer median survival (22.9 vs. 12.8 months, P = 0.02). The results of both phase 2 and 3 trials in the platinum-resistant treatment suggest that the benefit from BV is independent of chemotherapy partners, affording flexibility for physicians in choosing appropriate cytotoxic agents for treating patients in the salvage setting.
Ongoing Trials with Bevacizumab
Several other phase 3 trials of BV for advanced ovarian cancer are currently underway. The first of these, GOG 0213, seeks to clarify the role of BV in recurrent, platinum-sensitive disease in combination with chemotherapy (carboplatin plus either paclitaxel or docetaxel).(39) The primary objective of this trial is to determine whether a second round of surgical cytoreduction and adjuvant chemotherapy, with or without BV, improves overall survival. Another phase 3 trial, GOG 0252, compares the safety and efficacy of BV plus I.V. paclitaxel/carboplatin with BV plus intraperitoneal (I.P.) chemotherapy (paclitaxel plus either I.P. carboplatin or I.P. cisplatin) as first-line therapy.(40) This trial is no longer recruiting patients; the completion date is expected in December 2015. A different phase 3 study has been initiated to evaluate the safety profile associated with longer exposure to these agents while further investigating BV in combination with carboplatin and paclitaxel as therapy for patients with advanced ovarian cancer. The patients in this study will be exposed to combination therapy for up to a total of 8 cycles and BV for up to a total of 36 cycles. The secondary outcome measures for the study include PFS and overall response rate.(41) A German phase 3 trial (BOOST) has been initiated to evaluate the optimal duration of first-line chemotherapy in patients using BV in combination with paclitaxel and carboplatin (6 cycles).(42) Patients will be randomized to receive BV alone after combination therapy for either an additional 16 or 38 cycles until disease progression or unacceptable toxicity. A trial in the United Kingdom (OSCAR 1) will evaluate the safety and efficacy of bevacizumab in combination with chemotherapy as firstline treatment in patients with advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer in routine clinical practice.(43)
Multitargeted Antiangiogenic Agents
The small molecule receptor tyrosine kinase inhibitors (TKIs) are a major class of antiangiogenic agents. These agents pass freely through cell membranes and disrupt intracellular signaling by preventing the phosphorylation of VEGFR (1-3), PDGFR, c-Kit, and FGFR, and subsequent activation of downstream pathways (Please refer to the illustration on page 4).
In Phase III Clinical Development
The multitargeted antiangiogenic agents that are currently or have been in phase 3 clinical development for the treatment of advanced ovarian cancer include trebananib (AMG 386), nintedanib (BIBF 1120), and cediranib. Trebananib is an antiangiogenic peptide-Fc fusion protein (peptibody) that prevents interaction between the Tie2 receptor on endothelial cells and its ligands, angiopoietin1 and 2 (Ang1 and Ang2).(44) The Tie2 signaling activity promotes angiogenesis through increased endothelial cell survival, proliferation, and migration. Ang2 is consistently overexpressed at sites of active angiogenesis, and functions in a parallel proangiogenic pathway.(45,46)
Previous results from a randomized, placebo controlled phase 2 trial suggested that trebananib improved PFS in a dose-dependent manner.(47) Patients with recurrent ovarian cancer were randomized to receive paclitaxel (80 mg/m2 qw) in combination with trebananib 10 mg/kg qw, trebananib 3 mg/kg qw, or placebo qw. Although none of the efficacy measures reached statistical significance, patients treated with higher levels of trebananib had a substantially longer median PFS than those who received lower dose trebananib (7.2 vs. 5.7 months) or placebo (7.2 vs. 4.6 months). Additionally, the longer PFS associated with the higher dose trebananib treatment led to a survival benefit compared to the lower dose (22.5 vs. 20.4 months) or placebo (22.5 vs. 20.9 months). The ORR reported for this trial was 37%, 19%, and 27%, respectively. The CA125 responses were 71% and 28% for the investigational and placebo arms. After pharmacokinetic/pharmacodynamic analyses, investigators suggested the dose of 15 mg/kg of trebananib qw for future evaluation. A phase Ib trial that evaluated the efficacy of trebananib 15 mg/kg qw in combination with paclitaxel 175 mg/m2 q3w/carboplatin AUC 6 q3w as first-line therapy in women with high-risk early stage and advanced ovarian cancer found that the higher dose was tolerable in combination with cytotoxic chemotherapy.(48)
In response to the encouraging phase 2 data, several phase 3 trials (TRINOVA-1, TRINOVA-2, and TRINOVA-3) were planned to evaluate the efficacy of trebabanib; early results from TRINOVA-1 have been reported. TRINOVA-1 evaluated the efficacy of paclitaxel plus trebananib for women with recurrent, partially platinum-sensitive or resistant advanced EOC, PPC, or fallopian tube cancer in a randomized, placebo-controlled phase trial.(49) In the topline results, the researchers found a statistically significant difference in PFS with a 34% reduction in the risk of disease progression or death (HR = 0.66, 95 percent CI, 0.57, 0.77, P < 0.001). The median PFS was 7.2 months in the trebananib arm versus 5.4 months in the control arm. The data for the OS is expected in 2014.(50) The primary outcome measures from a second planned phase 3 trial, TRINOVA-2, evaluating the addition of trebananib to pegylated liposomal doxorubicin are expected in October 2014.(51) Finally, the TRINOVA-3 phase 3 trial will evaluate trebananib in combination with paclitaxel and carboplatin as the first-line therapy for advanced ovarian cancer.(52) The participants will be randomized to treatment with trebananib 15 mg/kg qw or placebo in combination with standard paclitaxel/carboplatin until progression or unacceptable toxicity. The primary outcome measure is PFS.
Nintedanib is a small molecule tyrosine kinase inhibitor with antiangiogenic activity due to its inhibition of VEGFR, PDGFR, and FGFR signaling.(53,54) The activity of this agent as maintenance therapy for recurrent ovarian cancer was investigated in a randomized, placebo controlled phase 2 study.(55) Participants who had responded to their most recent chemotherapy regimen were treated with nintedanib 250 mg twice daily or placebo continuously for 36 weeks. Greater than half of the trial participants entered onto the study after at least a third line of therapy. At the end of treatment, PFS rates were 16.3% and 5.0% for nintedanib- and placebo-treated patients, respectively. Although the trial was not powered for direct comparison of PFS or OS, the hazard ratio for the two groups was 0.65 and 0.84, respectively. An ongoing, large phase 3 trial, the AGO-OVAR 12 trial, was initiated to evaluate the role of nintedanib in combination with standard frontline chemotherapy in the treatment of advanced ovarian cancer.(56) Some preliminary results were presented at an international meeting in 2013; while the OS data is still immature, the researchers found that the PFS was significantly longer with nintedanib and paclitaxel/carboplatin than with placebo and paclitaxel/carboplatin (median 17.3 vs 16.6 months; HR 0.84; 95% CI:0.72 – 0.98; P = 0.0239).
Cediranib is an oral inhibitor of the 3 VEGF tyrosine kinases (VEGF- 1, 2, 3), thereby blocking VEGF signaling, angiogenesis, and tumor cell growth. While there had been a halt in development due to disappointing results with cediranib in metastatic colorectal cancer (57) and glioblastoma,(58) this agent may receive additional life because of the findings in ovarian cancer. Cediranib had shown some activity in ovarian cancer.(59,60) These findings spurred the initiation of the randomized, three-armed, double-blinded phase 3 ICON6 trial which evaluated the efficacy and safety of platinum-based chemotherapy in combination with cediranib in women with platinum-sensitive relapsed ovarian cancer.(61) Patients were randomly assigned to platinum-based chemotherapy/placebo with placebo maintenance therapy (reference arm), cediranib 20 mg/day during chemotherapy with placebo maintenance (concurrent arm), or cediranib 20 mg/day during chemotherapy with cediranib maintenance (maintenance arm). The treatment continued for 18 months or until disease progression and the chemotherapy consisted of up to six cycles of carboplatin/ paclitaxel, carboplatin/gemcitabine, or single-agent platinum therapy. PFS resulted in a more than three month advantage with cediranib and OS resulted in an approximately three month advantage that favored cediranib (restricted means analyses).(62) These findings suggest that since cediranib may be beneficial in ovarian cancer.
ENMD 2076 is a small molecule kinase inhibitor able to suppress angiogenesis and cell proliferation through the selective inhibition of Aurora A, VEGFR, FGFR, src, c-KIT, and FAK.(63) Preclinical studies have shown that ENMD 2076 was able to prevent the formation of new blood vessels and cause the regression of formed vessels in vivo. During a phase 1 trial of ENMD-2076 in advanced solid tumors, two patients with platinum-refractory ovarian cancer experienced partial response by RECIST criteria.(64) Based on these findings, an open-label phase 2 trial of ENMD-2076 was conducted in patients with recurrent/refractory ovarian cancer.65 Patients were initially treated with ENMD-2076 325 mg/d, but the dose was reduced to 275 mg/d due to toxicities and treatment delays.
Metronomic Chemotherapy
Another treatment strategy for ovarian cancer is the dosing of chemotherapeutic agents to low, repetitive levels that preferentially affect stromal and endothelial cells rather than tumor cells. Several studies of metronomic chemotherapy schedules have been conducted in patients with advanced recurrent ovarian cancer.(66)
A preclinical dose-finding study of topotecan in an orthotopic model of advanced ovarian cancer found that while the MTD of topotecan caused higher morbidity than metronomic topotecan, tumor vascularity was suppressed more in response to metronomic treatment.(67) Compared to controls, metronomic dosing yielded that greatest reduction in vascular density (32-33%, P < 0.01). Another preclinical study assessed the addition of pazopanib to topotecan and found combination therapy superior to metronomic topotecan alone in terms of suppression of tumor cell proliferation (P < 0.01).(68) The investigators have recommended pazopanib and metronomic topotecan for clinical development. After favorable results in a breast cancer trial that demonstrated activity of metronomic chemotherapy in heavily pretreated patients,(69) a phase 2 trial of BV 10 mg/kg q2w in combination with low dose metronomic cyclophosphamide 50 mg/d was conducted in patients with recurrent ovarian cancer.(70) The findings of this trial included a 56% 6-month PFS rate and a partial response rate of 24%. The median time to progression was 7.2 months and median survival was 16.9 months.
Biomarkers
The emergence of several antiangiogenic agents as efficacious in treating advanced ovarian cancer has encouraged researchers to identify biomarkers that will reveal the mechanisms of action of toxicity or activity. Identification of prognostic and predictive biomarkers that will direct treatment decisions represents the “brass ring” of clinical research in the era of targeted therapy. With the goal of improving efficacy and minimizing toxicity, selecting patients for therapy based on tumor biology and patient characteristics remains a challenge in the majority of tumor types. Recent research has identified the microRNA biomarker miR-181a which may help predict treatment response in epithelial ovarian cancer.(71) There are several other possible biomarkers for early detection of disease, prediction of treatment response, and overall prognosis.(72) To date, no reliable and specific biomarkers for angiogenesis and treatment with antiangiogenic agents have been identified. Despite the lack of validated biomarkers, clinical observations in ovarian cancer provide clues for these potential candidate biomarkers. As previously mentioned, high levels of tumor VEGF is associated with poorer prognosis in patients with advanced ovarian cancer.(73,74) The multivariate analysis of one study showed that VEGF expression in tumors, by immunohistochemistry, and disease stage were independent prognostic indicators of survival (P = 0.008 and P = 0.006, respectively).(75)
A recently reported study of the loss of heterozygocity (LOH) in the entire genome of ovarian tumors indicated that genomic instability in ovarian cancer is a significant and independent negative prognostic factor for both PFS and OS.(76) Investigators assessed the prevalence of LOH in numerous ovarian tumors using whole genome Single Nucleotide Polymorphism (SNP) arrays and found that the average percentage of the genome with LOH was 35%, and that the fraction of the genome with LOH correlated significantly with OS (P = 0.0000034) and PFS (P = 0.0016). This data, however, requires further validation and evaluation of its clinical utility.
Safety of Antiangiogenic Therapy for Ovarian Cancer
Antiangiogenic agents, relative to cytotoxic chemotherapy, are well tolerated and typically associated with fewer treatment interruptions and discontinuations due to toxicities. Nevertheless, VEGF is critical for the health and maintenance of a number of organ systems, and pharmacological disruption of its physiology is associated with a number of distinct class-effect adverse events that require careful monitoring and management. Among the most notable adverse effects associated with VEGF-targeted therapy are hypertension, arterial and venous thromboembolism, impaired wound healing, bleeding, and proteinuria.(77,78,79,80) These classeffect toxicities are related to the targeted action of antiangiogenic agents.
Bowel perforation is an unusual but potentially life-threatening complication of VEGF inhibition and appears to be slightly more prevalent among ovarian cancer patients than other tumor types. Variable rates of bowel perforation have been reported for studies of BV for recurrent ovarian cancer. A study of single-agent BV in recurrent ovarian cancer found a rate of GI bowel perforation of 11.4% (five of 44), which led to the early closure of the study.(81) All five patients had radiographic evidence of bowel involvement in disease at study initiation. The phase 3 trials of BV in patients with ovarian cancer have not produced similar rates of bowel perforation.
The GOG 0218 trial reported a rate of 5.4% for all grade > 2 GI events, which included perforation, fistula, necrosis or anastomotic leak.(82) The rates of GI perforation in BV-treated patients reported for the ICON7 and OCEANS trials were 1% and 0, respectively.(83,84) A retrospective review of 82 patients with recurrent epithelial ovarian cancer treated with BV at a single institution between 2006 and 2009 identified bowel perforation in 8 patients (9.8%).(85) Among these cases, a history of bowel surgeries (87.5% versus 24.3%; P = 0.0008) and the presence of symptoms of bowel obstruction (100% versus 18.9%; P ≤0.0001) were markedly higher in women who developed this complication than in those who did not. A separate retrosplysis identified 4 incidents cases of GI bowel perforations (7%) in 62 BV-treated patients, with all occurring in patients with large intra-abdominal tumor burdens who responded to BV treatment.(86) An additional study evaluated the risk factors for GI adverse events in a phase 3 trial of bevacizumab as a first-line therapy.(87) Out of the 1,759 (94%) that were evaluable, 2.8% (50 of 1,759) of the patients developed GI AEs. While the precise mechanisms remain unclear, one hypothesis posits that tumor necrosis and regression coupled with impairment of healing mechanisms during angiogenesis inhibition may weaken segments of the bowel wall already compromised by tumor and obstruction.(88) Univariate analyses of the phase 3 trial demonstrated that previous treatment of inflammatory bowel disease (P = 0.005), small bowel resection (P = 0.032), or large bowel resection (P = 0.012) at the time of primary surgery were significantly associated with a GI AE.(89) Patients with bowel involvement by tumor, bowel obstruction, and who have undergone heavy pretreatment (≥3 prior regimens) with cytotoxic chemotherapy may be particularly susceptible to bowel perforation during antiangiogenic therapy.(90)
Future Directions
A number of published clinical studies and reports have now validated the role of antiangiogenic therapeutics in the management of advanced ovarian cancer. Both GOG 0218 and ICON7 established that the addition of BV to first-line standard chemotherapy improves PFS in this type of tumor. Ongoing studies such as GOG 0252 are also evaluating if adding BV to different routes and schedules for administering standard cytotoxic chemotherapy (for example, I.P. and dose-dense chemotherapy) can build upon the developments in both disease-free and overall survival. Future clinical evaluations of BV should include defining the optimal duration of BV therapy, both pre-and post-progression, and assessing the efficacy and safety of monotherapy with an anti-VEGF agent between chemotherapy courses. As new agents representing different components of the angiogenesis process undergo clinical development, the ideal sequencing of agents in the treatment of ovarian cancer must be considered. The identification of clinically validated biomarkers will aid in stratifying patients based on their likelihood to respond to angiogenesis inhibitors as well as direct treatment decision across multiple lines of therapy.
| Study | Setting | Treatment | ORR | mPFS | mOS |
|---|---|---|---|---|---|
| Phase 316 (GOG-0218) |
First-line therapy of advanced ROC |
paclitaxel/carboplatin: +PL +BV 15 mg/kg q3w initial +BV 15 mg/kg q3w throughout |
— | 10.3 months 11.2 months 14.1 months (P < 0.001)* |
39.3 months 38.7 months 39.7 months* (P = 0.45)* |
| Phase 317 (ICON7) |
First-line therapy of high-risk, early stage, and advanced ROC |
paclitaxel/carboplatin: alone +BV 7.5 mg/kg q3w |
48% 67% (P < 0.001) |
17.4 months 19.8 months (P = 0.04) |
44.6 months19 45.5 months (P = 0.85) |
| Phase 317 (ICON7) |
First-line therapy of high-risk, early stage, and advanced ROC |
paclitaxel/carboplatin: alone +BV 7.5 mg/kg q3w |
48% 67% (P < 0.001) |
17.4 months 19.8 months (P = 0.04) |
44.6 months19 45.5 months (P = 0.85) |
| Phase 320 (OCEANS) |
Recurrent, platinumsensitive ROC |
Gemcitabine/carboplatin: +PL +BV 15 mg/kg q3w to PD |
57.4% 78.5% (P < 0.0001) |
8.4 months 12.4 months (P < 0.0001) |
32.9 months23 33.6 months |
| Phase 325 (AURELIA) |
Platinum resistant ROC | Chemo (investigators choice): paclitaxel, topotecan or PLD alone +BV 15 mg/kg q3w |
11.8% 27.3% (P = 0.0 01) |
3.4 months 6.7 months (P < 0.001) |
13.3 months 16.6 months NS |
| Phase 349,50 (AGO-OVAR 16) |
Second-line therapy of FIGO stage II-IV EOC, PPC |
Pazopanib 800 mg/day | — | — | HR = 1.076 |
| Phase 338 (TRINOVA-1) |
Recurrent, partially platinum-sensitive or resistant advanced EOC, PPC |
Paclitaxel/PL Paclitaxel + trebananib 15 mg/kg qw |
— | 5.4 months 7.2 months P < 0.001 |
NR (2014) |
| Phase 345 (AGO-OVAR 12) |
Advanced ovarian cancer | PL+ paclitaxel/carboplatin nintedanib + paclitaxel/carboplatin |
17.3 months 16.6 months P = 0.0239 |
17.3 months 16.6 months P = 0.0239 |
— |
| Phase 356 (ICON6) |
Platinum-sensitive relapsed ovarian cancer |
Platinum-based chemotherapy + PL/PL maintenance cediranib 20 mg/day + chemotherapy/ PL cediranib 20 mg/day + chemotherapy/maintenance cediranib |
3 month advantage favoring cediranib |
3 month advantage favoring cediranib |
|
| Phase 222 | Recurrent, platinumrefractory EOC PPC |
BV 15 mg/kg q3w 21% | 4.7 months | 17 months | |
| Phase 213 | Platinum-resistant EOC PSC |
BV 15 mg/kg q3w | 15.9% | 4.4 months | 10.7 months |
| Phase 281 | Recurrent, platinumrefractory EOC PPC |
Metronomic cyclophosphamide + BV 10 mg/kg q2w |
24% | 7.2 months (TTP) |
16.9 months |
| Phase 215 | First-line/adjuvant therapy of ROC or PSMC |
Carboplatin/paclitaxel + BV 15 mg/kg q3w |
75% | 29.8 months | NR |
| Phase 226 | Recurrent, platinum-resistant EOC PPC |
nab-paclitaxel + BV 15 mg/kg q2w | 46.1% | 8.3 months | 16.5 months |
| Phase 236 | Recurrent ROC | Paclitaxel + trebananib 10 mg/kg qw trebananib 3 mg/kg qw PL qw |
37% 19% 27% |
7.2 months* 5.7 months 4.6 months (P = 0.225)* |
22.5 months* 20.4 months 20.9 months |
| Phase 244 | Relapsed ROC | Nintedanib 250 mg twice daily or PL 250 mg twice daily |
— | 16.3 % 5% (PFS rate at 36 wks, P = .06) |
( P = .51) |
| Phase 248 GEICO | Recurrent ROC | Pazopanib 800 mg daily to PD | 4% | 1.8 months | |
| Phase 247 | Recurrent ROC | Pazopanib 800 mg daily to PD | 18% | 6-month PFS 17% |
— |
| Phase 268 | Platinum-resistant ROC | Sunitinib 50 mg daily 4 of 6 weeks, or Sunitinib 37.5 mg daily continuously |
16.7% 5.4% (P > .11) |
4.8 months 2.9 months (P = .3048) |
13.6 months 13.7 months (P = .8380) |
| Phase 259 | Recurrent, platinumresistant ROC with malignant ascites |
Aflibercept 4 mg/kg q2w | — | 59.5 days | 92 days |
ORR, objective response rate; mPFS, median progression-free survival; mOS, median overall survival; ROC, epithelial ovarian, primary peritoneal and fallopian tube cancer; PL, placebo; BV, bevacizumab; NR, not reported/reached; PD, progressive disease; PLD, pegylated liposomal doxorubicin; EOC, epithelial ovarian cancer; PSC, peritoneal serous cancer; PPC, primary peritoneal cancer; PSMC, papillary serous mullerian carcinoma.
* as compared with the control group
**data not yet mature
REFERENCES
1. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9-29.
2. American Cancer Society. Cancer Facts and Figures 2014. Atlanta, GA: American Cancer Society; 2014.
3. American Cancer Society. Cancer Facts and Figures 2014. Atlanta, GA: American Cancer Society; 2014.
4. Folkman J, Watson K, Ingber D, Hanahan D. Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature. 1989;339:58-61.
5. Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. Jun 2003;9:669-676.
6. Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. Jun 2003;9:669-676.
7. Jain RK. Molecular regulation of vessel maturation. Nat Med. Jun 2003;9(6):685-693.
8. Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. Jun 2003;9:669-676.
9. Ferrara N. Molecular and biological properties of vascular endothelial growth factor. J Mol Med (Berl). 1999;77(7):527-543
10. Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. Jun 2003;9:669-676.
11. Ramakrishnan S, Subramanian IV, Yokoyama Y, Geller M. Angiogenesis in normal and neoplastic ovaries. Angiogenesis. 2005;8(2):169-182.
12. Ramakrishnan S, Subramanian IV, Yokoyama Y, Geller M. Angiogenesis in normal and neoplastic ovaries. Angiogenesis. 2005;8(2):169-182.
13. Shen GH, Ghazizadeh M, Kawanami O, et al. Prognostic significance of vascular endothelial growth factor expression in human ovarian carcinoma. Br J Cancer. Jul 2000;83(2):196-203.
14. Dvorak HF. Vascular permeability factor/vascular endothelial growth factor: a critical cytokine in tumor angiogenesis and a potential target for diagnosis and therapy. J Clin Oncol. Nov 1 2002;20(21):4368-4380.
15. Byrne AT, Ross L, Holash J, et al. Vascular endothelial growth factortrap decreases tumor burden, inhibits ascites, and causes dramatic vascular remodeling in an ovarian cancer model. Clin Cancer Res. Nov 15 2003;9(15):5721-5728.
16. Byrne AT, Ross L, Holash J, et al. Vascular endothelial growth factortrap decreases tumor burden, inhibits ascites, and causes dramatic vascular remodeling in an ovarian cancer model. Clin Cancer Res. Nov 15 2003;9(15):5721-5728.
17. Avastin [package insert]. South San Francisco, CA: Genentech, Inc.; 2013.
18. Burger RA. Experience with bevacizumab in the management of epithelial ovarian cancer. J Clin Oncol. Jul 10 2007;25(20):2902-2908.
19. Cannistra SA, Matulonis UA, Penson RT, et al. Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J Clin Oncol. Nov 20 2007;25(33):5180-5186.
20. Micha J, Goldstein B, Rettenmaier M, et al. A phase II study of outpatient first-line paclitaxel, carboplatin, and bevacizumab for advanced-stage epithelial ovarian, peritoneal, and fallopian tube cancer. Int J Gynecol Cancer. 2007;17: 771–776.
21. Penson RT, Dizon DS, Cannistra SA, et al. Phase II study of carboplatin, paclitaxel, and bevacizumab with maintenance bevacizumab as firstline chemotherapy for advanced mullerian tumors. J Clin Oncol. Jan 1 2010;28(1):154-159.
22. Micha J, Goldstein B, Rettenmaier M, et al. A phase II study of outpatient first-line paclitaxel, carboplatin, and bevacizumab for advanced-stage epithelial ovarian, peritoneal, and fallopian tube cancer. Int J Gynecol Cancer. 2007;17: 771–776.
23. Penson RT, Dizon DS, Cannistra SA, et al. Phase II study of carboplatin, paclitaxel, and bevacizumab with maintenance bevacizumab as firstline chemotherapy for advanced mullerian tumors. J Clin Oncol. Jan 1 2010;28(1):154-159.
24. Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. Dec 29 2011;365(26):2473-2483.
25. Perren TJ, Swart AM, Pfisterer J, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. Dec 29 2011;365(26):2484-2496.
26. Stark D, Nankivell M, Pujade-Lauraine E et al. Standard chemotherapy with or without bevacizumab in advanced ovarian cancer: quality-of-life outcomes from the International Collaboration on Ovarian Neoplasms (ICON7) phase 3 randomised trial. The Lancet Oncology. 2013;14 :236-243.
27. Oza AM Perren TJ, Swart AM et al. ICON7: Final overall survival results in the GCIG phase III randomized trial of bevacizumab in women with newly diagnosed ovarian cancer. European Cancer Congress 2013. Abstract 6.
28. Aghajanian C, Blank SV, Goff BA, et al. OCEANS: A Randomized, Double- Blind, Placebo-Controlled Phase III Trial of Chemotherapy With or Without Bevacizumab in Patients With Platinum-Sensitive Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer. J Clin Oncol. Jun 10 2012;30(17):2039-2045.
29. Pfisterer J, Plante M, Vergote I, et al. Gemcitabine plus carboplatin compared with carboplatin in patients with platinum-sensitive recurrent ovarian cancer: an intergroup trial of the AGOOVAR, the NCIC CTG, and the EORTC GCG. J Clin Oncol. Oct 10 2006;24(29):4699-4707.
30. Burger RA, Sill MW, Monk BJ, Greer BE, Sorosky JI. Phase II trial of bevacizumab in persistent or recurrent epithelial ovarian cancer or primary peritoneal cancer: a Gynecologic Oncology Group Study. J Clin Oncol. Nov 20 2007;25(33):5165-5171.
31. Aghajanian C, Blank SV, Goff BA, et al. OCEANS: A Randomized, Double- Blind, Placebo-Controlled Phase III Trial of Chemotherapy With or Without Bevacizumab in Patients With Platinum-Sensitive Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer. J Clin Oncol. Jun 10 2012;30(17):2039-2045.
32. Aghajanian C, Goff BA, Nycum LR et al. Final analysis of overall survival in OCEANS, a randomized phase III trial of gemcitabine, carboplatin, and bevacizumab followed by bevacizumab until disease progression in patients with platinum-sensitive recurrent ovarian cancer. 2014 Society of Gynecologic Oncology 45th Annual Meeting on Women’s Cancer March 22 – March 25, 2014. Abstract 137.
33. Pujade-Lauraine, Eric and Hilpert, Felix and Weber, Béatrice et al. Bevacizumab Combined With Chemotherapy for Platinum-Resistant Recurrent Ovarian Cancer: The AURELIA Open-Label Randomized Phase III Trial. JCO. 2013:51:4489; published online on March 17, 2014.
34. Naumann RW, Coleman RL: Management strategies for recurrent platinumresistant ovarian cancer. Drugs. 2011;71:1397-1412.
35. Tillmanns TD, Lowe MP, Schwartzberg LS, et al. Phase II clinical trial of bevacizumab with albumin-bound paclitaxel in patients with recurrent, platinum-resistant primary epithelial ovarian or primary peritoneal carcinoma. Gynecologic Oncology. 2013;128: 221–228.
36. McGonigle KF, Muntz HG, Vuky J, et al. Combined weekly topotecan and biweekly bevacizumab in women with platinum-resistant ovarian, peritoneal, or fallopian tube cancer: results of a phase 2 study. Cancer. Aug 15 2011;117(16):3731-3740.
37. Tillmanns TD, Lowe MP, Schwartzberg LS, et al. Phase II clinical trial of bevacizumab with albumin-bound paclitaxel in patients with recurrent, platinum-resistant primary epithelial ovarian or primary peritoneal carcinoma. Gynecologic Oncology. 2013;128: 221–228.
38. McGonigle KF, Muntz HG, Vuky J, et al. Combined weekly topotecan and biweekly bevacizumab in women with platinum-resistant ovarian, peritoneal, or fallopian tube cancer: results of a phase 2 study. Cancer. Aug 15 2011;117(16):3731-3740.
39. Carboplatin and Paclitaxel With or Without Bevacizumab After Surgeryin Treating Patients With Recurrent Ovarian Epithelial Cancer, Primary Peritoneal Cavity Cancer, or Fallopian Tube Cancer. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 3]. Available from: http://clinicaltrials.gov/ct2/show/NCT00565851 NLM Identifier: NCT00565851.
40. Bevacizumab and Intravenous or Intraperitoneal Chemotherapy in Treating Patients With Stage II, Stage III, or Stage IV Ovarian Epithelial Cancer, Fallopian Tube Cancer, or Primary Peritoneal Cancer. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 3] Available from: http://clinicaltrials.gov/ct2/show/NCT00951496 NLM Identifier:NCT00951496.
41. A Study of the Addition of Avastin (Bevacizumab) to Carboplatin and Paclitaxel Therapy in Patients With Ovarian Cancer. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000-[cited 2014 May 3]. Available from: http://clinicaltrials.gov/ct2/show/NCT01239732 NLM Identifier: NCT01239732.
42. Evaluation of Optimal Initial Treatment Duration of Bevacizumab in Combination With Standard Chemotherapy in Patients With Ovarian Cancer (BOOST). In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 3]. Available from: http://clinicaltrials.gov/ ct2/show/NCT01462890 NLM Identifier:NCT01462890.
43. An Observational Study of Avastin (Bevacizumab) in Combination With Chemotherapy as First-Line Treatment in Patients With Advanced Ovarian Cancer (OSCAR 1). In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 3]. Available from: http://clinicaltrials.gov/ show/NCT01863693 NLM Identifier: NCT01863693
44. Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand SJ, Holash J. Vascular-specific growth factors and blood vessel formation. Nature. 2000;407(6801):242-248.
45. Zhang L, Yang N, Park JW, et al. Tumor-derived vascular endothelial growth factor up-regulates angiopoietin-2 in host endothelium and destabilizes host vasculature, supporting angiogenesis in ovarian cancer. Cancer Res. Jun 15 2003;63(12):3403-3412.
46. Holash J, Maisonpierre PC, Compton D, et al. Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science. Jun 18 1999;284(5422):1994-1998.
47. Karlan BY, Oza AM, Richardson GE, et al. Randomized, double-blind, placebocontrolled phase II study of AMG 386 combined with weekly paclitaxel in patients with recurrent ovarian cancer. J Clin Oncol. Feb 1 2012;30(4):362-371
48. Casado A, Oaknin A, Baurain J-F, et al. Phase Ib study of AMG 386 in combination with paclitaxel (P) and carboplatin (C) in high-risk stage I and stages II-IV epithelial ovarian, primary peritoneal, or fallopian tube cancers. ASCO Meeting Abstracts. May 30, 2012 2012;30(15_suppl):5038.
49. TRINOVA-1: A Study of AMG 386 or Placebo, in Combination With Weekly Paclitaxel Chemotherapy, as Treatment for Ovarian Cancer, Primary Peritoneal Cancer and Fallopian Tube Cancer. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US).2000- [cited 2014 May 6]. Available from: http://clinicaltrials.gov/ct2/show/NCT01204749 NLM Identifier: NCT01204749.
50. Amgen Press Release. Available at http://www.amgen.com/media/media_pr_ detail.jsp?year=2013&releaseID=1829205. Accessed on May 5, 2014.
51. TRINOVA-2: TRial IN OVArian Cancer-2. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 6]. Available from: http://clinicaltrials.gov/ct2/show/NCT01281254. NLM Identifier: NCT01281254.
52. TRINOVA-3: A Study of AMG 386 or AMG 386 Placebo in Combination With Paclitaxel and Carboplatin to Treat Ovarian Cancer. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 6]. Available from: http://clinicaltrials.gov/ct2/show/NCT01493505. NLM Identifier: NCT01493505.
53. Erber R, Thurnher A, Katsen AD, et al. Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. Faseb J. Feb 2004;18(2):338-340.
54. Hilberg F, Roth GJ, Krssak M, et al. BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. Jun 15 2008;68(12):4774-4782.
55. Ledermann JA, Hackshaw A, Kaye S, et al. Randomized phase II placebocontrolled trial of maintenance therapy using the oral triple angiokinase inhibitor BIBF 1120 after chemotherapy for relapse ovarian cancer. J Clin Oncol. Oct 1 2011;29(28):3798-3804.
56. LUME-Ovar 1: Nintedanib (BIBF 1120) or Placebo in Combination With Paclitaxel and Carboplatin in First Line Treatment of Ovarian Cancer. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2014 May 6]. Available from: http://clinicaltrials.gov/ct2/show/ NCT01015118 NLM Identifier: NCT01015118.
57. Hans-Joachim Schmoll, David Cunningham, Alberto Sobrero et al. ediranib With mFOLFOX6 Versus Bevacizumab With mFOLFOX6 As First-Line Treatment for Patients With Advanced Colorectal Cancer: A Double-Blind, Randomized Phase III Study (HORIZON III). JCO. 2012;42:5355.
58. Tracy T. Batchelor, Paul Mulholland, Bart Neyns et al. Phase III Randomized Trial Comparing the Efficacy of Cediranib As Monotherapy, and in Combination With Lomustine, Versus Lomustine Alone in Patients With Recurrent Glioblastoma. JCO 2012;47:2464.
59. Hirte HWV, Fleming GF, Sugimoto AK et al. A phase II study of cediranib (AZD2171) in recurrent or persistent ovarian, peritoneal or fallopian tube cancer: final results of a PMH, Chicago and California consortia trial. J Clin Oncol. 2008. Abstract 5521.
60. Matulonis UA, Berlin S, Ivy P et al. Cediranib, an oral inhibitor of vascular endothelial growth factor receptor kinases, is an active drug in recurrent epithelial ovarian, fallopian tube, and peritoneal cancer. J Clin Oncol. 2009; 27:5601–5606.
61. ICON6 Trial website. Available at http://www.icon6.org/about_icon6/ overview.aspx
62. Ledermann JA, Perren TJ, Raja FA et al. Randomised double-blind phase III trial of cediranib (AZD 2171) in relapsed platinum sensitive ovarian cancer: Results of the ICON6 trial. European Cancer Congress 2013 (ECCO-ESMO-ESTRO): Abstract LBA 10. Presented September 30, 2013.
63. Fletcher GC, Brokx RD, Denny TA, et al. ENMD-2076 is an orally active kinase inhibitor with antiangiogenic and antiproliferative mechanisms of action. Mol Cancer Ther. Jan 2011;10(1):126-137.
64. Diamond JR, Bastos BR, Hansen RJ, et al. Phase I safety, pharmacokinetic, and pharmacodynamic study of ENMD-2076, a novel angiogenic and Aurora kinase inhibitor, in patients with advanced solid tumors. Clin Cancer Res. Feb 15 2011;17(4):849-860.
65. Matulonis U, Tew WP, Matei D, Behbakht K, Fleming GF, Oza AM. A phase II study of ENMD-2076 in platinum-resistant ovarian cancer. ASCO Meeting Abstracts. June 9, 2011 2011;29(15_suppl):5021.
66. Shen GH, Ghazizadeh M, Kawanami O, et al. Prognostic significance of vascular endothelial growth factor expression in human ovarian carcinoma. Br J Cancer. Jul 2000;83(2):196-203.
67. Merritt WM, Danes CG, Shahzad MM, et al. Anti-angiogenic properties of metronomic topotecan in ovarian carcinoma. Cancer Biol Ther. Aug 2009;8(16):1596-1603.
68. Merritt WM, Nick AM, Carroll AR, et al. Bridging the gap between cytotoxic and biologic therapy with metronomic topotecan and pazopanib in ovarian cancer. Mol Cancer Ther. Apr 2010;9(4):985-995.
69. Colleoni M, Rocca A, Sandri MT, et al. Low-dose oral methotrexate and cyclophosphamide in metastatic breast cancer: antitumor activity and correlation with vascular endothelial growth factor levels. Ann Oncol. Jan 2002;13(1):73- 80.
70. Garcia AA, Hirte H, Fleming G, et al. Phase II clinical trial of bevacizumab and low-dose metronomic oral cyclophosphamide in recurrent ovarian cancer: a trial of the California, Chicago, and Princess Margaret Hospital phase II consortia. J Clin Oncol. Jan 1 2008;26(1):76-82.
71. Aditya Parikh, Christine Lee, Peronne Joseph et al. microRNA-181a has a critical role in ovarian cancer progression through the regulation of the epithelial–mesenchymal transition. Nature Communications, 2014; 5.
72. Sreeja Sarojini, Ayala Tamir, Heejin Lim, et al. Early Detection Biomarkers for Ovarian Cancer. Journal of Oncology. 2012; doi:10.1155/2012/709049.
73. Ramakrishnan S, Subramanian IV, Yokoyama Y, Geller M. Angiogenesis in normal and neoplastic ovaries. Angiogenesis. 2005;8(2):169-182.
74. Shen GH, Ghazizadeh M, Kawanami O, et al. Prognostic significance of vascular endothelial growth factor expression in human ovarian carcinoma. Br J Cancer. Jul 2000;83(2):196-203.
75. Shen GH, Ghazizadeh M, Kawanami O, et al. Prognostic significance of vascular endothelial growth factor expression in human ovarian carcinoma. Br J Cancer. Jul 2000;83(2):196-203.
76. Korpanty G, Timms K, Abkevich V, et al. Loss of heterozygosity (LOH) as a measure of whole-genome instability in ovarian cancer correlates with clinical outcomes. ASCO Meeting Abstracts. June 9, 2011; 29(15_suppl):5027.
77. Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. Dec 29 2011;365(26):2473-2483.
78. Aghajanian C, Blank SV, Goff BA, et al. OCEANS: A Randomized, Double- Blind, Placebo-Controlled Phase III Trial of Chemotherapy With or Without Bevacizumab in Patients With Platinum-Sensitive Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer. J Clin Oncol. Jun 10 2012;30(17):2039-2045.
79. Gressett SM, Shah SR. Intricacies of bevacizumab-induced toxicities and their management. Ann Pharmacother. Mar 2009;43(3):490-501.
80. Hurwitz HI, Saltz LB, Van Cutsem E, et al. Venous thromboembolic events with chemotherapy plus bevacizumab: a pooled analysis of patients in randomized phase II and III studies. J Clin Oncol. May 1 2011;29(13):1757-1764.
81. Cannistra SA, Matulonis UA, Penson RT, et al. Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J Clin Oncol. Nov 20 2007;25(33):5180-5186.
82. Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. Dec 29 2011;365(26):2473-2483.
83. Perren TJ, Swart AM, Pfisterer J, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. Dec 29 2011;365(26):2484-2496.
84. Aghajanian C, Blank SV, Goff BA, et al. OCEANS: A Randomized, Double- Blind, Placebo-Controlled Phase III Trial of Chemotherapy With or Without Bevacizumab in Patients With Platinum-Sensitive Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer. J Clin Oncol. Jun 10 2012;30(17):2039-2045.
85. Tanyi JL, McCann G, Hagemann AR, et al. Clinical predictors of bevacizumabassociated gastrointestinal perforation. Gynecol Oncol. Mar 2011;120(3):464- 469.
86. Wright JD, Secord AA, Numnum TM, et al. A multi-institutional evaluation of factors predictive of toxicity and efficacy of bevacizumab for recurrent ovarian cancer. Int J Gynecol Cancer. May-Jun 2008;18(3):400-406.
87. Robert A. Burger, Mark F. Brady, Michael A. Bookman et al. Risk Factors for GI Adverse Events in a Phase III Randomized Trial of Bevacizumab in First-Line Therapy of Advanced Ovarian Cancer: A Gynecologic Oncology Group Study. JCO. Apr 20, 2014:1210-1217; published online on March 17, 2014.
88. Wright JD, Secord AA, Numnum TM, et al. A multi-institutional evaluation of factors predictive of toxicity and efficacy of bevacizumab for recurrent ovarian cancer. Int J Gynecol Cancer. May-Jun 2008;18(3):400-406.
89. Wright JD, Secord AA, Numnum TM, et al. A multi-institutional evaluation of factors predictive of toxicity and efficacy of bevacizumab for recurrent ovarian cancer. Int J Gynecol Cancer. May-Jun 2008;18(3):400-406.
90. Tanyi JL, McCann G, Hagemann AR, et al. Clinical predictors of bevacizumabassociated gastrointestinal perforation. Gynecol Oncol. Mar 2011;120(3):464- 469.
CME REQUIREMENTS
Copyright 2014 by The Angiogenesis Foundation. All rights reserved.
ACCREDITATION STATEMENT
This activity has been planned and implemented by Ciné-Med and the Angiogenesis Foundation Ciné-Med is accredited by the American Nurses Credentialing Center (ANCC), the Accreditation Council for Pharmacy Education (ACPE), and the Accreditation Council for Continuing Medical Education (ACCME), to provide continuing medical education for the health care team.
CREDIT DESIGNATION
Ciné-Med designates this enduring activity for a maximum of 1 AMA PRA Category 1 Credit(s)™. Physicians should only claim credit commensurate with the extent of their participation in the activity. Credit will be awarded to those who complete this activity according to the instructions and achieve a score of 70% or better on the post activity examination. A certificate of credit will be available for download to those who successfully complete the examination.
RELEASE AND EXPIRATION
Date of original release: June 24, 2014
Date of expiration: June 23, 2015
CME Course Code: 2014OVARIAN
TARGET AUDIENCE
Practicing oncologists and primary care physicians in the U.S., researchers and medical students
HEALTHCARE GAP
In the United States alone, more than 22,240 new cases of ovarian cancer are estimated to have been diagnosed in 2013, and an estimated 14,230 will have died from this disease. Ovarian cancer is the ninth most common cancer among women, excluding non-melanoma skin cancers. It ranks fifth in cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system. Ovarian cancer accounts for about 3% of all cancers in women. A woman’s risk of getting ovarian cancer during her lifetime is about 1 in 72. Her lifetime chance of dying from ovarian cancer is about 1 in 100.
PROGRAM LEARNING OBJECTIVES
At the completion of this activity, participants should be able to:
• Summarize the current clinical progress of targeted therapies in the management of ovarian cancer in early and late stage clinical trials.
• Explain current clinical challenges in the management of ovarian cancer.
• Interpret the outcome data from recent well-designed scientific and clinical studies of protocols studying new-targeted therapies.
• Integrate side effect management into the long term management of patients with ovarian cancer.
• Assess treatment options, efficacy data, and side effect management with members of the cancer treatment team, as well as cancer patients and their family members.
ACTIVITY GOAL
This activity is designed to address the following ABMS / IOM competencies: Patient Care and Medical Knowledge
METHOD OF PARTICIPATION
There are no fees for participating in and receiving credit for this online educational activity. The participant should, in order, read the objectives and faculty disclosures, review the educational content, answer the multiple-choice post-test and complete the evaluation. This program is available in PDF format accessible from the Angiogenesis Foundation’s website (http://www.angio.org) in the CME section. A print version is also available; for more information contact outreach@angio.org. After reviewing the material, CME credits are available through the Angiogenesis Foundation’s website (http://www.cmeonline.org) by selecting the name of the program (registration required). Course code: 2014OVARIAN
ACKNOWLEDGEMENT OF SUPPORT
This activity is supported by educational grants from Genentech.
COURSE FACULTY
William Li, MD Dartmouth Medical College
DISCLOSURE
Cine-Med adheres to accreditation requirements regarding industry support of continuing medical education. Disclosure of the planning committee and faculty’s commercial relationships will be made known at the activity. Speakers are required to openly disclose any limitations of data and/or any discussion of any offlabel, experimental, or investigational uses of drugs or devices in their presentations. All Cine-Med and Angiogenesis Foundation employees in control of content have indicated that they have no relevant financial relationships to disclose.
William W. Li, M.D. President, the Angiogenesis Foundation Editor-in-Chief
Disclosure: No relevant financial relationships to disclose.
TOPICS AND EDUCATIONAL CONTENT
The State of Antiangiogenic Therapy for Advanced Ovarian Cancer:
• The State of Antiangiogenic Therapy for Advanced Ovarian Cancer
• Anti-VEGF Monoclonal Antibody • Multitargeted Antiangiogenic Agents
• Metronomic Chemotherapy
• Biomarkers
• Safety of Antiangiogenic Therapy for Ovarian Cancer
• Future Directions
SYSTEM REQUIREMENTS
This educational program is available as a mobile application and a PDF file. The mobile application can be run on any iOS or Android device. To view and print PDF files, you must have Adobe Reader installed on your computer. Most computers already have this software installed. If yours does not, you can download Adobe Reader free from the Adobe Web site: http://www.adobe.com. For questions about this program, please contact the Angiogenesis Foundation at 617-401-2779 or outreach@angio.org.